Nice presentation and graphics. The talk will please general science literate audience. However, the technical results from results could be explained a bit more for more technical audience. Particulalry the bar graphs that you show in one of the slides was not addressed or explained in the talk. It went by too fast. Overall, it is a great presentation. Best Wishes.
-
-

Dear Jonathan: I enjoyed your presentation. It seems like you have a large number of variables with which to control ESC behaviors. My question is “what particularly is the novelty” of your work in a crowded field of stem cell bioengineers?
Thanks, and best wishes.
-
Hey Professor Moghe,
Thanks for your interest in my work.
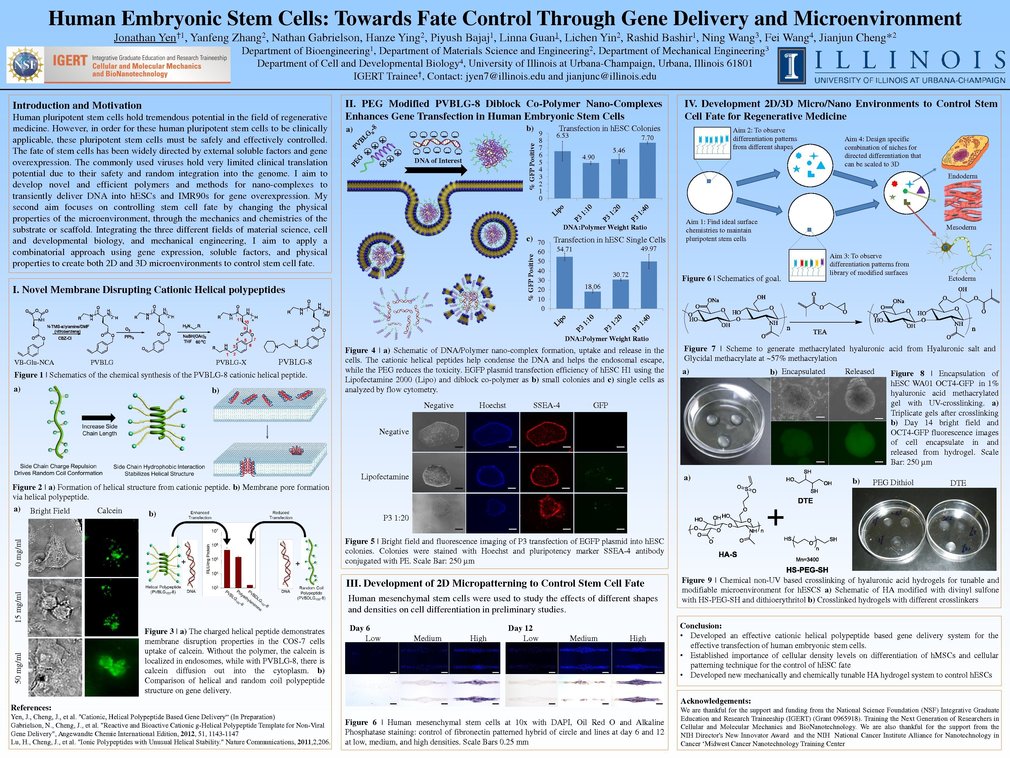
Recent work using 3D scaffolds for stem cell culture have focused on mesenchymal stem cells. While this is an important area of research, the clinical utility of mesenchymal stem cells is limited in comparison to that of embyronic stem cells. In my study, I seek to build upon our current understanding of 3D culturing techniques and specifically apply it to embryonic stem cell culture. This increased clinical utility also raises certain challenges to overcome. The pluripotent nature of embryonic stem cells increases the possibilities of cell fate, and comprehensive studies must be done in order to define conditions that exist in the maintenance and differentiation of these cells. So many engineering groups have been looking at individual variables at a time, from mechanical cues to adhesion peptides. Currently many methods require the use embryoid bodies to differentiate human embryonic stem cells into certain cell lines. But the efficiency of these embryoid body derived cells is low and results in many other differentiated cell lineages, which makes purification quite tedious. Therefore there are more and more approaches for directed differentiation in 2D of hESCs. But these 2D established neurons or neural progenitor cells are grown on rigid petri dishes which in turn can be hard to maintain these cells long in culture and maintain their phenotypes. Neurons have been found to favor extremely soft and specific microenvironment in the body. My 3D culturing system aims to serve as a platform for structuring niches. This hydrogel system is customizable, and thus can suit a diversity of cell differentiation applications. Of these possibilities, our lab focuses on the neuro-ectoderm differentiation stage of embryonic stem cells. In this, we seek to define optimal conditions for the growth and development of neurons or neural progenitor cells that could be used as regenerative therapeutics in instances of neurological disease.
Thanks!
Jonathan Yen -
very nice and interesting talk and poster.
It seems as though the development of your platform technology is progressing well. But where do you set your sights in terms of testing feasibility? What is the specific end application? Is it blood vessels, liver, heart, etc? Based on this, what would be the issues one faces in terms of customizing your technology to a specific end goal? I think the lack of a solid objective leaves these questions open.
-
Hi Professor Shin,
Thank you for your interest. These are important questions. Overall, the 3D system I am developing ideally would be fully tunable and functionalizable, so that when new cues or microenvironments are identified for specific differentiation, it can be incorporated or tuned accordingly. But currently my main goals/target is to be able to differentiate human embryonic stem cells in neuroectoderm lineages. Therefore I plan to move towards spinal cord injury repair. Our lab has already achieved highly efficient neuro conversion in 2D, but they are not specific to any specific neurons. Therefore through the use of microenvironments, I am working towards a 3D microenvironment to further direct and develop hESCs into specific neurons (motor or dopaminergic neurons) that can be maintained and expanded in this 3D culture for downstream analysis. I believe that the large issue I would face would be the isolation and purification of the differentiated neurons to obtain high purity for in-vivo applications. In addition, the ultimate goal would be to generate a specific 3D scaffold to efficiently (~90%) differentiate hESCs into neurons, in which they can directly be incorporated in-vivo for spinal repair. But this would also assume that there are no residual pluripotent stem cells remaining that can cause teratomas.
Thanks!
Jonathan Yen -
Hi Jonathan, your work seems pretty useful, and you do a pretty fantastic job of explaining how you’ve achieved your current level of progress in such a short time. Your opening slides discussed a very broad range of potential applications, which opens up some big doors. In the same vein as Prof. Shin above, do you have any particular target applications/collaborators in mind?
-
Hey Erich!
Thanks for the support!
As in my response to Professor Shin above, I am looking into neuro differentiation specifically for spinal repair. But this is still in the early stages, and we are working first on the differentiation aspect of the cells. In the future we do hope to conduct in-vivo studies to see the functionalities of the neurons. I believe that through the IGERT CMMB program and UIUC, it has given me opportunities to interact with other researchers and thus allow for potential future collaborations.Thanks
Jonathan Yen -
Further posting is closed as the event has ended.





Judges and Presenters may log in to read queries and replies.