Using Nature to Guide Materials Synthesis: Hierarchically Structured Inorganic-Inorganic Nanocomposites Formed in Silica Hydrogel
Alternative energy systems are currently limited by the trade-offs between mutually exclusive sets of properties. For example, more efficient solar cells require nanostructuring, which often come at a cost to the electron mobility. Single crystal materials with nanocomposite structures embody the potential to optimize such related properties independently.
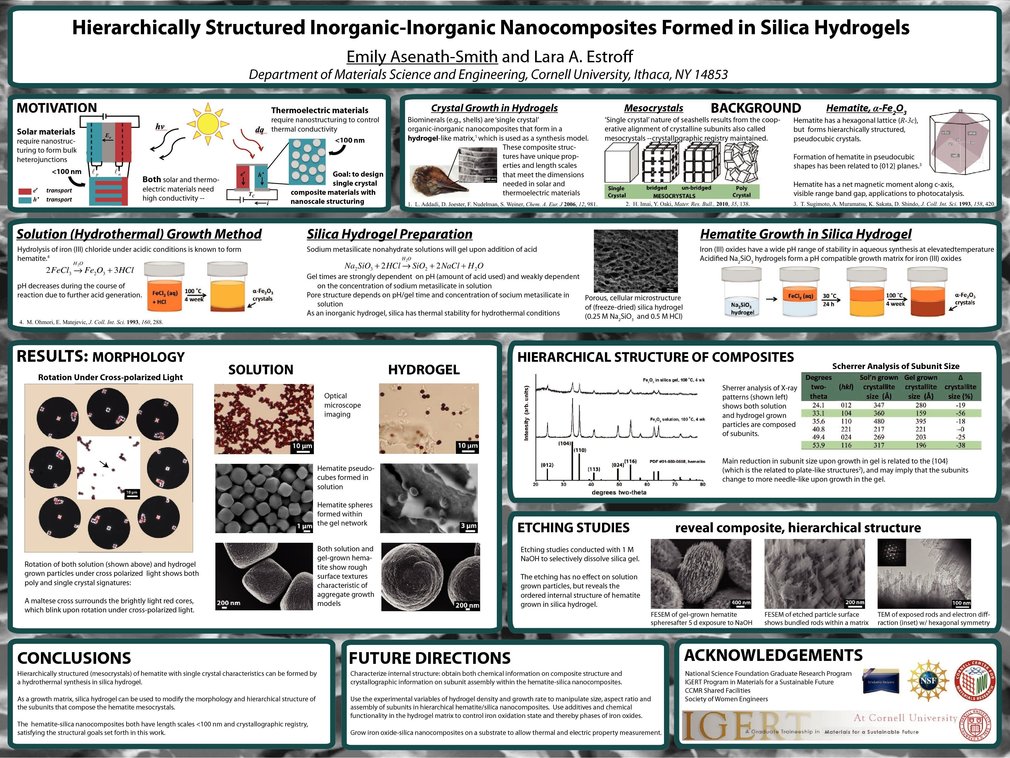
Synthesis models for crystalline materials abound in Nature and include the formation of (bio)minerals by living organisms in a gel-like matrix of biopolymers. Many biominerals, such as seashells, exhibit single crystal characteristics due to hierarchical structuring, but are also composite materials due to the presence of incorporated biopolymers. In this work silica hydrogel is used as a bio-inspired growth matrix for the formation of hierarchically structured a-Fe2O3 (hematite) nanocomposites.
Polarized light microscopy, X-ray diffraction, scanning electron microscopy, and transmission electron microscopy were used to characterize the iron oxide particles. Based upon these results, the hematite phase was confirmed and the hierarchical structure was revealed in both solution and gel grown crystals. The ability to manipulate the aspect ratio of the subunits by growth in a hydrogel was demonstrated. Etching experiments revealed the gel-grown hematite particles to contain incorporated silica, qualifying them as inorganic-inorganic composites. While the solution grown crystals were stable upon exposure to etching in base, the gel grown particles underwent partial dissolution, revealing an ordered internal structured composed of bundled rods with crystallographic registry and dimensions <100 nm. These length scales, composite nature and crystallographic registry are in agreement with the design criteria for alternative energy systems.



Judges and Presenters may log in to read queries and replies.